


BUILDING A CONNECTED ECOSYSTEM FOR SAFER SURGERY

A perioperative medication safety platform that standardizes preparation, documentation, and waste—without slowing the OR.
In the operating room, medication preparation and labeling have historically relied on manual steps that vary by person and setting—each introducing risk. Variation at preparation can lead to vial and ampoule swaps, or handwritten/illegible syringe labels; at administration, it can drive syringe swaps—three well-recognized medication errors hospitals work hard to prevent. Meanwhile, documentation by the anesthesia provider is often completed under time pressure or after the fact, and controlled-substance waste and witnessing typically occur later, creating blind spots in reconciliation and oversight.
Safety hinges on in-workflow verification and standardization—across preparation, administration, and waste. Verify a medication’s identity and concentration at preparation, carry that identity forward with a standardized, barcode label at administration, and capture/reconcile waste at the point of care. Without these embedded checks, variability persists case-to-case and room-to-room, and preventable errors and incomplete or delayed documentation follow.
The result is a fragmented landscape with wide variability in medication use. Practices can differ between hospitals—and even between operating rooms in the same facility—making consistent safety enforcement difficult. Despite efforts to strengthen protocols, perioperative medication management can function like an open loop, leaving persistent error patterns that technology must address inside the workflow.
Codonics saw these risks and set out to change the game.
What began as a company known for its precision labeling technology has now evolved into a global medication safety platform that spans the entire perioperative process from drug preparation and administration to waste and reconciliation.
Today, Codonics’ innovative solutions close the safety loop by creating a fully connected ecosystem, ensuring traceability at every step and transforming the OR into a safer, more efficient environment.
The OR is one of the most dynamic environments in healthcare, and we’ve designed our systems to fit that environment perfectly–compact, intuitive, interoperable, and built to enhance—not interrupt—care.
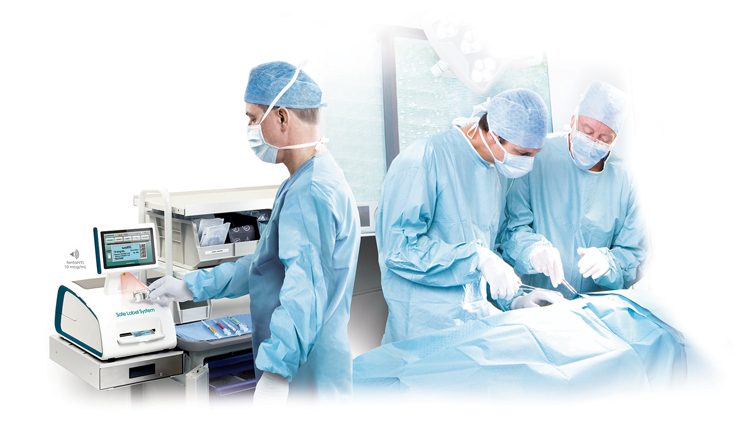
The Label That Sparked a Revolution
For Codonics, it all started with a label but not just any label. It began with the recognition that labeling was the most error-prone and least standardized aspect of perioperative medication use. In a world that relied heavily on handwritten identifiers and ad-hoc processes, Codonics envisioned a future where medication safety was embedded directly into workflow, not bolted on after the fact.
That vision became the Safe Label System® (SLS), an “FDA-cleared” Class II medication safety device and first-in-its-class solution for the operating room. Long before the Institute for Safe Medication Practices (ISMP) or The Joint Commission (TJC) issued formal guidance, Codonics had already built a system to anticipate those very standards. The company standardized the high-risk steps of medication preparation and labeling, long before “perioperative safety” was a global conversation.

As Peter O. Botten, president and CEO reflects, “Codonics Safe Label System turned medication safety guidelines into best practice. It standardizes the high-risk steps, adds safety, strengthens documentation and supports 340B-related accountability, and helps clinicians move faster with confidence across thousands of ORs worldwide.”
The Safe Label System became the foundation of a larger movement that aligned technology, pharmacy, and clinical culture into a seamless safety net for anesthesia care.
Building a Platform for Safer Surgery
While SLS solved the long-standing problem of mislabeling, Codonics didn’t stop there. Over time, it grew into a platform that redefined how safety, efficiency, and compliance coexisted in the OR.
At the core of this evolution lies a philosophy: standardization without slowdown. Codonics’ technology doesn’t ask clinicians to work differently. It simply makes their existing workflow safer, faster, and traceable. By bridging automation and human expertise, the platform ensures that every drug prepared, administered, and disposed of in the OR is identified, measured, assayed, witnessed, and reconciled.
Today, the Codonics ecosystem includes SLS for safety, verification and compliant labeling, SLS-WAVE® for handsfree documentation into leading AIMS/EHR systems, and the upcoming Safe Waste System™ (SWS), a first-of-its-kind innovation that redefines controlled substance waste, assay, and reconciliation at the point of care.
“We’ve always believed that technology should elevate people by improving their workflows,” says Botten. “The OR is one of the most dynamic environments in healthcare, and we’ve designed our systems to fit that environment perfectly–compact, intuitive, interoperable, and built to enhance—not interrupt—care.”
From Preparation to Waste
Medication safety doesn’t end once a syringe is filled. It extends to every step of the medication lifecycle, from preparation to administration to waste. That full-circle perspective defines Codonics’ next chapter.
The Safe Waste System (SWS) represents a transformative leap forward. Using built-in spectrophotometer technology, SWS enables anesthesia providers to identify, measure, witness, waste, assay, and reconcile controlled substance waste, in <7 seconds, and all at the point of care. What was once a multi-step, multi-department process now occurs in real-time in the OR— faster, more transparent, reducing diversion risk, strengthening pharmacy oversight, and easing administrative burden. It’s a win-win.
“This is the natural evolution of what we started,” Botten explains. “SLS standardized preparation, SLS-WAVE transformed administration, and now SWS will redefine waste and reconciliation. Together the Codonics platform creates a single, connected safety ecosystem that supports every provider and every patient.”
Built to Integrate not Interrupt
Codonics’ success lies not only in what its technology does, but how it does it. Designed for integration rather than disruption, the system works seamlessly with leading automated and manual anesthesia drug carts, the BD Intelliport® system, interoperable syringe pumps, and AIMS/EHR systems like Epic and Cerner.
When clinicians log into an automated cart that’s integrated with Safe Label System, it logs the user into both systems. A single scan of a vial or ampoule verifies vial barcode data and provides visual and audible cues for drug and concentration, then prints a full-color, TJC-compliant syringe label in seconds. The syringe label barcode enables hands-free electronic documentation at administration via SLS-WAVE and adds another layer of safety; dose entry remains manual unless Intelliport® is used.
The result is fewer manual clicks, better charge capture, and more accurate patient records, all without adding time or complexity to clinical workflows.
At several world-class academic medical centers and top ranked children’s hospital, such as Texas Children’s Hospital, Codonics’ SLS helped resolve a labeling citation and later earned recognition from surveyors as a “best practice.” At UVA Health and the University of Washington, labeling compliance climbed to nearly 100 percent, with improved documentation integrity and greater clinician confidence. These case examples underscore Codonics’ core philosophy: measurable outcomes, not just technological promise.
Anticipating the Future of Safety
The Codonics roadmap is designed to stay ahead of guidance, not chase it. The company’s innovations have consistently aligned with the Anesthesia Patient Safety Foundation’s (APSF) STPC paradigm—Standardization, Technology, Pharmacy/Prefilled/Premixed, and Culture—well before it became a benchmark.
The next phase focuses on RFID-enabled traceability, expanding the barcode-driven workflows that have become standard across Codonics installations. The addition of highfrequency (HF) and ultra-high-frequency (UHF) RFID read/write capability to syringe labels and containers is set to further strengthen medication tracking, compliance, and audit readiness.
Codonics Safe Label System turned medication safety guidelines into best practice. It standardizes the high-risk steps, adds safety, strengthens documentation and supports 340B-related accountability, and helps clinicians move faster with confidence across thousands of ORs worldwide.
Data-driven insights are another key frontier. By analyzing barcode and RFID data across the enterprise, hospitals can identify predictive risk signals, monitor workflow bottlenecks, and maintain real-time dashboards that elevate safety oversight from reactive to proactive.
Codonics’ vision is clear: the OR of the future will not only be safer, it will be smarter.
The Human Side of Innovation
At its core, Codonics is a partnership company. Every implementation is guided by the Codonics Implementation Team (CIT), and together with a group of clinical, pharmacy, and engineering experts, ensure that technology adoption translates to lasting practice.
From workflow mapping and network reviews, to live training and post-launch optimization, the aim is simple: make the “safe thing the easy thing–and sustain it,” as Botten puts it. The company’s emphasis on collaboration ensures that hospitals achieve both compliance and clinician buy-in, turning technology into trusted practice.
The team’s ability to bridge Pharmacy, Anesthesiology, IT, and BioMed has made Codonics the partner of choice for enterprise deployments across the U.S. and abroad. The result is a rare combination of clinical safety, operational efficiency, and organizational alignment.
Global Recognition, Local Impact
Today, Codonics is a standard of care, supporting 16,000+ operating rooms across multiple countries and enabling hundreds of millions of medication preparations. In reported implementations, hospitals have seen up to 95% reductions in medication errors and 30% efficiency gains, depending on local workflow and scope.
Named a Top 100 HealthTech Company, Codonics has earned multiple awards, including Medical Design Excellence Awards (MDEA) and recognitions for best practices in medication safety.
But behind those accolades lies a deeper mission—one that continues to guide every product release and partnership.
As Botten emphasizes, “Our mission is to make medication safety second nature. The goal is not just compliance, it’s confidence. We want every clinician to know that the system supporting them is as precise and dependable as they are.”
The Future Is Connected
Looking ahead, Codonics’ roadmap points to even broader horizons. Beyond SWS and RFID, the company is investing in deeper Epic and Cerner interoperability, barcode-to-pump workflows, and real-time analytics dashboards. The goal is to transform data into intelligence, helping hospitals see not just what went wrong, but what might.
As healthcare continues to evolve, Codonics remains anchored in the principle that defined its beginning: that safety and speed are not opposites, but allies. Its technology strengthens care.
What began with a label has become a connected, closed loop medication management platform that helps standardize medication safety from preparation to waste. For clinicians, it’s support they can feel at the point of care. For patients, it’s a safeguard they rarely see but always benefit from.
And for Codonics, it’s the foundation for what comes next.
Regulatory note: Codonics Safe Label System (SLS) is an “FDA-cleared” Class II medical device intended for automatic verification of drug identity using barcode data from NDC and other drug vial UDI barcodes. It enables creation of ISO 26825/ASTM 4774 color-coded labels in alignment with TJC-MM and ISMP guidance. Caution: Federal law restricts this device to sale by or on the order of a licensed practitioner.

